Number of views:
1000
Rifampicin Capsules (Ⅱ)
Retail price
0.0
元
Market price
0.0
元
Number of views:
1000
Product serial number
Quantity
-
+
Stock:
Category
1
Product description
Parameters
Rifampicin Capsules (Ⅱ) Instructions
Please read the instructions carefully and use under the guidance of a doctor
【Drug Name】
Generic name: Rifampicin Capsule (Ⅱ)
English name: Rifampicin Capsules (Ⅱ)
Chinese Pinyin: Lipuping Jiaonang (Ⅱ)
【Ingredients】
Chemical name: 3-[[(4-methyl-1-piperazinyl)imino]methyl]rifamycin.
Chemical Structure:
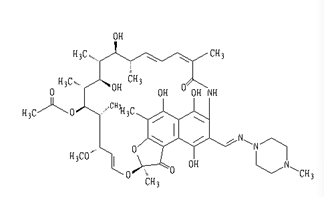
Molecular formula: C 43 H 58 N 4 O 12
Molecular weight: 822.95
【Properties】
This product is a capsule, and the content is a purple pansy waxy solid.
【Indications】
1. This product is used in combination with other anti-tuberculosis drugs for the initial treatment and retreatment of various tuberculosis, including the treatment of tuberculous meningitis.
2. This product is used in combination with other drugs for the treatment of leprosy and non-tuberculous mycobacterial infections.
3. This product and vancomycin (intravenous) can be combined for serious infections caused by methicillin-resistant Staphylococcus. The combined regimen of rifampicin and erythromycin is used for serious infections of the genus Legionella.
4. For asymptomatic carriers of Neisseria meningitidis to eliminate Neisseria meningitidis in the nasopharyngeal area; but not suitable for the treatment of Neisseria meningitidis infection.
【specification】
225mg (calculated as C 43 H 58 N 4 O 12 )
【Dosage】
oral.
0.45g (2 capsules) once a day or as directed by a doctor.
【Adverse reactions】
1. Gastrointestinal reactions are the most common. After oral administration of this product, gastrointestinal reactions such as anorexia, nausea, vomiting, upper abdominal discomfort, and diarrhea may occur. The incidence rate is 1.7% to 4.0%, but all of them can be tolerated.
2. Liver toxicity is the main adverse reaction of this product, with an incidence of about 1%. In the first few weeks of the treatment course, a small number of patients may have elevated serum aminotransferase, hepatomegaly and jaundice, most of which are asymptomatic transient elevated serum aminotransferase, which can recover on their own during the course of treatment. The elderly and alcoholics , Malnutrition, pre-existing liver disease or other factors causing abnormal liver function are more likely to occur.
3. "Flu-like syndrome" may occasionally appear after high-dose intermittent therapy for allergies, which manifests as chills, chills, fever, discomfort, dyspnea, dizziness, lethargy and muscle pain, etc. The frequency of occurrence, dose size, and interval time may vary. Obviously related. Acute hemolysis or renal failure may occasionally occur, and it is currently believed that the mechanism is an allergic reaction.
4. After other patients take this product, urine, saliva, sputum, tears, etc. may appear orange-red. Occasionally, leukopenia, shortened prothrombin time, headache, dizziness, visual disturbance, etc.
【Taboo】
1. People who are allergic to this product or rifamycin antibacterial drugs should not be used.
2. Patients with severe liver dysfunction, biliary obstruction and pregnant women within 3 months are disabled.
【Precautions】
1. Use with caution in people with alcoholism and liver damage. Use with caution in infants, pregnant women over 3 months and breastfeeding women.
2. Interference with diagnosis: it can cause direct antiglobulin test (Coombs test) to be positive; interfere with the determination of serum folic acid concentration and serum vitamin B12 concentration; it can cause false positives in the sulfobromide sodium test; it can interfere with the use of spectrophotometry The results of various urinalysis tests carried out due to changes in meter or color; can make blood urea nitrogen, serum alkaline phosphatase, serum alanine aminotransferase, aspartate aminotransferase, serum bilirubin, and serum urea nitrogen, serum alkaline phosphatase, serum alanine aminotransferase, aspartate aminotransferase, serum bilirubin, and serum urea nitrogen, serum alkaline phosphatase, serum alanine aminotransferase, aspartate aminotransferase, serum bilirubin, and serum urea nitrogen, serum alkaline phosphatase, serum alanine aminotransferase, aspartate aminotransferase, serum bilirubin, and serum The measurement result of uric acid concentration increased.
3. Rifampicin can cause liver insufficiency. There are reports of deaths with jaundice in patients with original liver disease or when this product is taken with other hepatotoxic drugs. Therefore, patients with original liver disease can only be treated if there are clear indications. Use with caution. Observe the changes in liver function before and during treatment. Once liver damage occurs, stop the drug immediately.
4. Hyperbilirubinemia is a mixed type of hepatocellularity and bile retention. Mild patients resolve spontaneously during medication, and severe patients need to be discontinued for observation. Elevated blood bilirubin may also be the result of competition between rifampicin and bilirubin for excretion. Liver function changes should be closely monitored during the initial 2 to 3 months of treatment.
5. When rifampicin is used alone to treat tuberculosis or other bacterial infections, pathogens can quickly develop drug resistance, so this product must be used in combination with other drugs. Treatment may take 6 months to 2 years, or even several years.
6. Rifampicin may cause a decrease in white blood cells and platelets, and cause bleeding and infection of the gums, and delayed wound healing. At this time, you should avoid surgery such as tooth extraction, and pay attention to oral hygiene, brush your teeth and floss your teeth carefully, until the blood picture returns to normal. During the medication, the surrounding blood should be checked regularly.
7. Rifampicin should be taken 1 hour before a meal or 2 hours after a meal. It is best absorbed once in the morning on an empty stomach, because eating affects the absorption of this product.
8. Patients with impaired liver function often need to reduce the dose, and the daily dose is ≤8mg/kg.
9. People with impaired renal function do not need to reduce the dose. There was no significant change in the blood concentration of rifampicin in patients with reduced glomerular filtration rate or anuria.
10. After taking the medicine, urine, saliva, sweat and other excrement can be orange-red. There is a possibility of interstitial nephritis.
[Medicine for pregnant women and lactating women]
1. Rifampicin can pass through the placenta, animal experiments have caused teratogenesis. Although there is no report of teratogenicity in humans, there is not enough data to show that it can be used safely in pregnancy. It is forbidden for pregnant women within 3 months. Use with caution in pregnant women over 3 months.
2. Rifampicin can be excreted in breast milk. Women who take the drug during lactation should fully weigh the pros and cons before deciding whether to use the drug.
【Children's Medication】
The safety of this product in children under 5 years of age has not been established. Use with caution for babies.
【Geriatric Medication】
The liver function of elderly patients has decreased, so the dosage should be reduced accordingly.
【medicine interactions】
1. Drinking alcohol can increase the incidence of rifampicin-induced hepatotoxicity and increase the metabolism of rifampicin. It is necessary to adjust the dose of rifampicin and closely observe whether the patient has hepatotoxicity.
2. The aminosalicylate can affect the absorption of this product and cause its blood concentration to decrease; if it must be used in combination, the two should be taken at least 6 hours apart.
3. The combined use of this product and isoniazid may increase the risk of hepatotoxicity, especially in patients with original liver damage and rapid isoniazid acetylation.
4. The combination of rifampicin and ethionamide can aggravate its adverse reactions.
5. Chlorophenazine can reduce the absorption of rifampicin, delay the peak time and prolong the half-life.
6. Combining rifampicin with miconazole or ketoconazole can reduce the plasma concentration of the latter two, so this product should not be combined with imidazoles.
7. Adrenal corticosteroids (glucocorticoids, mineralocorticoids), anticoagulants, aminophylline, theophylline, chloramphenicol, clofibrate, cyclosporine, verapamil (verapamil), proper Carney, propafenone, trimethoprim, coumarin or indandione derivatives, oral hypoglycemic agents, corticotropin, dapsone, digitalis glycosides, disopyramide, quinidine, etc. When rifampicin is used in combination, the latter induces liver microsomal enzyme activity, which can weaken the efficacy of the above drugs. Therefore, in addition to digoxin and dapsone, the doses of the above drugs need to be adjusted before and during the course of treatment with rifampicin. . When this product is combined with coumarin or indandione, the prothrombin time should be measured daily or regularly, and the dosage should be adjusted accordingly.
8. This product can promote the metabolism of estrogen or reduce its enterohepatic circulation, reduce the effect of oral contraceptives, and cause irregular menstruation, inter-menstrual bleeding and unplanned pregnancy. Therefore, when taking rifampin, patients should switch to other contraceptive methods.
9. This product can induce liver microsomal enzymes, increase the metabolism of anti-tumor drugs dacarbazine and cyclophosphamide, form alkylated metabolites, and promote the reduction of white blood cells, so the dosage needs to be adjusted.
10. The combination of this product and diazepam (Valium) can increase the elimination of the latter and reduce the blood concentration, so the dosage needs to be adjusted.
11. This product can increase the metabolism of phenytoin sodium in the liver, so when the two are used together, the blood concentration of phenytoin sodium should be measured and the dosage should be adjusted.
12. This product can increase the degradation of levothyroxine in the liver, so the dose of levothyroxine should be increased when the two are used together.
13. This product can also increase the metabolism of methadone and mexiletine in the liver, causing methadone withdrawal symptoms and a decrease in the blood concentration of mexiletine. Therefore, the latter two need to be adjusted when used in combination.
14. Probenecid can compete with this product to be taken up by hepatocytes, increasing the blood concentration of this product and causing toxic reactions. But the effect is unstable, so probenecid is usually not suitable to increase the blood concentration of this product.
【Overdose】
1. Excessive performance:
Mental retardation; edema around the eyes or face; whole body itching; red man syndrome (skin mucous membranes and sclera are red or orange). People with primary liver disease, alcoholics or those taking other hepatotoxic drugs may cause death.
2. Treatment:
(1) Stop the drug.
(2) Gastric lavage, because patients often have nausea and vomiting, it is not suitable to induce vomiting; activated carbon paste is given after gastric lavage to absorb the residual rifampicin in the gastrointestinal tract; antiemetics are given to patients with severe nausea and vomiting.
(3) Intravenous fluids and diuretics are given to promote the excretion of drugs.
(4) Symptomatic and supportive therapy.
【Pharmacology and Toxicology】
Rifampicin is a semi-synthetic broad-spectrum antibacterial drug of the rifamycin class, which has antibacterial activity against a variety of pathogenic microorganisms. The drug has obvious bactericidal effects on Mycobacterium tuberculosis and some non-tuberculous mycobacteria (including Mycobacterium leprae, etc.) both inside and outside the host cell. Rifampicin has a good antibacterial effect on aerobic Gram-positive bacteria, including enzyme-producing strains of Staphylococcus and methicillin-resistant strains, Streptococcus pneumoniae, other Streptococcus, Enterococcus, Listeria, Bacillus anthracis, Perfringens, diphtheria, anaerobic cocci, etc. It also has high antibacterial activity against aerobic gram-negative bacteria such as Neisseria meningitidis, Haemophilus influenzae, and Neisseria gonorrhoeae. Rifampicin also has a good effect on Legionella, and has an inhibitory effect on pathogens such as Chlamydia trachomatis, lymphogranuloma venereal disease and psittacosis.
Bacteria have cross-resistance to rifamycin antibiotics. Rifampicin binds firmly to the β subunit of DNA-dependent RNA polymerase, inhibits the synthesis of bacterial RNA and prevents the enzyme from connecting with DNA, thereby blocking the RNA transcription process and stopping the synthesis of DNA and protein.
【Pharmacokinetics】
Rifampicin is well absorbed orally, and the plasma concentration reaches its peak 1.5 to 4 hours after taking the drug. The peak plasma concentration (Cmax) of 600 mg in adults is 7-9 mg/L, and the peak plasma concentration (Cmax) is 11 mg/L in children 6 months to 5 years old. This product is well distributed in most tissues and body fluids, including cerebrospinal fluid. When the meninges are inflamed, the drug concentration in the cerebrospinal fluid increases; it can also reach effective therapeutic concentrations in saliva; this product can pass through the placenta. The apparent volume of distribution (Vd) is 1.6L/kg. The protein binding rate is 80% to 91%. Taking the drug after eating can reduce the absorption of the drug by 30%. The blood elimination half-life (t1/2β) of the drug is 3 to 5 hours, which is shortened after multiple administrations, which is 2 to 3 hours. This product can be rapidly deacetylated by the action of self-induced microsomal oxidase in the liver to become the metabolite 25-desacetylrifampicin with antibacterial activity. After hydrolysis, the inactive metabolites are excreted in the urine.
This product is mainly excreted through the gall bladder and intestines, and can enter the enterohepatic circulation, but its deacetylated active metabolites have no enterohepatic circulation. 60% to 65% of the dose is excreted in feces, 6% to 15% of the drug is excreted in the original form, 15% is the active metabolite, and 7% is excreted in the inactive 3-formyl derivative. It can also be excreted through breast milk. In patients with impaired renal function, this product does not accumulate; due to the effect of self-induced liver microsomal oxidase, the excretion rate increases after 6-10 days of taking rifampicin; after high doses, the biliary excretion reaches saturation. Excretion may be delayed. Rifampicin cannot be cleared by hemodialysis or peritoneal dialysis.
【Storage】
Seal and store in a cool, dry place (no more than 20°C away from light).
【package】
(1) Aluminum-plastic blister packaging, 10 tablets/plate; carton packaging, 1 plate/box.
(2) Aluminum-plastic blister packaging, 10 tablets/plate; carton packaging, 2 plates/box.
(3) Aluminum-plastic blister packaging, 10 tablets/plate; carton packaging, 3 plates/box.
(4) Aluminum-plastic blister packaging, 10 tablets/plate; carton packaging, 6 plates/box.
【Validity Period】
Tentative 18 months
【Executive Standard】
State Food and Drug Administration National Drug Standard WS-10001-(HD-1551)-2005-2011
【Approval Number】
National Medicine Standard H21023360
【manufacturer】
Company Name: Shenyang Hongqi Pharmaceutical Co., Ltd.
Scan the QR code to read on your phone
Previous
Rifampicin Capsules (Ⅱ)
相关产品


Copyright hongqipharma.com All rights reserved Powered by:300.cn Shenyang 辽icp备12005917号-1 (辽)-非经营性-2018-0012
Add:Shenyang Hunnan New District envelope 6th Street Tel:024-23786268-8012 Fax:024-23786263