Instructions for capreomycin sulfate for injection
Please read the instructions carefully and use it under the guidance of a physician.
【Drug Name】
Generic name: capreomycin sulfate for injection
English name: Capreomycin Sulfate for Injection
Chinese Pinyin: Zhusheyong Liusuan Juanqumeisu
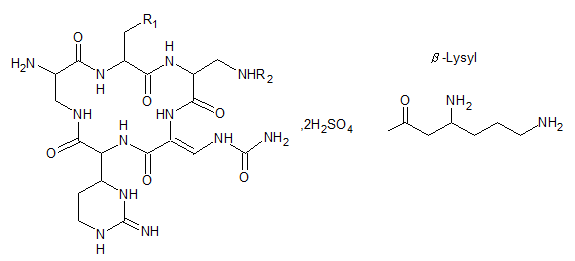
[Ingredients] The main ingredient of this product is capreomycin sulfate without auxiliary materials. The components include capreomycin IA, IB, IIA, and IIB. The sum of capreomycin IA and IB shall not be less than 90.0%; the sum of capreomycin IA, IB, IIA, and IIB shall not be less than 95.0%.
Chemical Structure:
Molecular formula:
Capreomycin IA: R1= OH, R2= β-Lysyl, C 25 H 44 N 14 O 8 Molecular weight: 668.71
Capreomycin IB: R1= H, R2= β-Lysyl, C 25 H 44 N 14 O 7 Molecular weight: 652.71
Capreomycin IIA: R1= OH, R2= H, C 19 H 31 N 12 O 7 Molecular weight: 539.51
Capreomycin II B: R1= H, R2= H, C 19 H 31 N 12 O 6 Molecular weight: 523.51
[Properties] This product is white or off-white powder; odorless; moisture-absorbing.
【Indications】
Applicable to second-line treatment drugs for tuberculosis, who have failed treatment with first-line anti-tuberculosis drugs (such as streptomycin, isoniazid, rifampicin and ethambutol), or have toxic effects on one or more of the above drugs Or when the bacteria are resistant, this product can be used as one of the combination drugs.
[Specifications] (1) 0.75g (750,000 units) (2) 1.0g (1 million units)
[Usage and Dosage] Take the medicine once a day for 2 to 4 months, then change to 2 to 3 times a week.
Intramuscular injection: 0.75~1g per day, once administered, add sterile water for injection to dissolve it immediately before use, and deep intramuscular injection.
Intravenous drip: 1g per day (body weight <55kg, 0.75g per day) once a day, dilute with 250ml sodium chloride injection before use and instill it, 60 drops/min. The total daily dose shall not exceed 20mg/kg.
Or follow the doctor's advice.
【Adverse reactions】
1. Intramuscular injection causes local pain and induration. It has been reported that bleeding or aseptic abscess at the injection site is found, and deep injection is recommended.
2. Transient blood pressure drops during intravenous injection, local phlebitis, low potassium, low calcium, low magnesium, electrolytes should be checked.
3. Renal function damage: BUN is slightly elevated, but high doses can still cause kidney tissue damage, such as hematuria, decreased urine output, decreased appetite, extreme thirst, etc., which should be regarded as important side effects and should be paid attention to.
4. Hearing disorders: tinnitus, ear fullness, hearing loss, unsteady gait, dizziness. It is not recommended to combine with parenteral anti-tuberculosis drugs (streptomycin, puromycin) and non-anti-tuberculosis drugs (polymyxin A sulfate, colistin sulfate, amikacin, etc.) with potential ototoxic or nephrotoxicity at the same time. Gentamicin, tobramycin, vancomycin, kanamycin, and neomycin) may be used in combination, or the risk of additional cranial nerve VIII damage or kidney damage should be carefully weighed by the treatment.
5. Electrolyte disorders: due to renal tubular damage, reduced K+ and Mg++ reabsorption, low potassium and low magnesium, manifested as weakness, lethargy, weak pulse, arrhythmia, dyspnea, abdominal distension, nausea, vomiting, and low calcium, may be Intramuscular convulsions and spasms.
6. A small number of cases, especially when capreomycin sulfate and other anti-tuberculosis drugs are administered at the same time, may have drug eruption and may be accompanied by drug fever.
7. Occasionally have symptoms such as arrhythmia, mental changes, headaches, etc.
8. Hemocytosis and leukopenia can be seen. Most patients treated with capreomycin sulfate daily have an increase in eosinophils of more than 5%. When the dose is reduced to 2-3g per week, the symptoms disappear. Thrombocytopenia is rarely reported.
[Taboo] People who are allergic to any of the ingredients in this product and pregnant women are prohibited.
【Precautions】
1. Patients with hearing loss, myasthenia gravis, Parkinson's disease, and renal insufficiency should be used with caution.
2. Pay attention to check before and during medication.
(1) Audiometry is done 1 to 2 times a week, preferably for electrical audiometry, once a month.
(2) The vestibular function and renal function are measured regularly. BUN and serum creatinine are slightly elevated in a large number of patients receiving long-term treatment, and most of these cases have found urine casts, red blood cells and white blood cells. Especially for patients with impaired renal function or the eighth pair of cranial neuropathy, 1-2 times a week, such as blood urea nitrogen 30mg/100ml or more or any other patients with decreased renal function with or without elevated BUN levels need to reduce or stop medicine.
(3) Liver function determination, especially when combined with other hepatotoxic anti-tuberculosis drugs.
(4) Electrolyte determination: As hypokalemia, hypomagnesemia and hypocalcemia may occur during treatment, the plasma levels of these electrolytes should be monitored in time. Especially before the medication and during the treatment, the blood potassium concentration should be measured once a month.
3. Interference with diagnosis: The results of phenolsulfonphthalein and sodium sulfobromophthalein excretion test are reduced; the measured values of blood urea nitrogen and non-protein nitrogen may increase.
4. In patients with dehydration, due to increased blood drug concentration, toxicity may increase.
5. When this product is used alone, bacteria can quickly develop drug resistance, so this product can only be used in combination with other antibacterial drugs for the treatment of tuberculosis. This product has cross-resistance with kanamycin and puromycin, but has no cross-resistance with other anti-tuberculosis drugs.
6. For patients with impaired renal function, adjust the dose according to the following table:
|
Creatinine clearance rate/(ml/min)/(ml/sec)
|
Dosage (calculated on base)
|
|
≥110 (1.84)
|
According to normal human consumption
|
|
100 (1.67)
|
1 time a day, 12.7mg/kg
|
|
80 (1.33)
|
Once a day, 10.4mg/kg
|
|
60 (1.00)
|
Once a day, 8.2mg/kg
|
|
50 (0.83)
|
7mg/kg daily, or 14mg/kg every 48 hours
|
|
40 (0.67)
|
5.9mg/kg daily, or 11.7mg/kg every 48 hours
|
|
30 (0.50)
|
4.7mg/kg daily, or 9.5mg/kg every 48 hours
|
|
20 (0.33)
|
3.6mg/kg daily, or 7.2mg/kg every 48 hours
|
|
10 (0.17)
|
2.4mg/kg daily, or 4.9mg/kg every 48 hours
|
|
0 (0)
|
1.3mg/kg daily, or 2.6mg/kg every 48 hours or 3.9mg/kg every 72 hours
|
7. If the condition improves after 2 to 3 weeks of medication, you still need to continue the entire course of treatment.
8. Deep intramuscular injection is required when injecting this product. Shallow injection can aggravate pain and cause aseptic abscess.
9. During the use of this product, if any adverse events and/or adverse reactions occur, please consult your doctor.
10. Please inform your doctor if you are using other medicines at the same time.
11. Please place it out of the reach of children.
12. Local neuromuscular blockade may occur after large-dose intravenous injection of capreomycin sulfate. This effect was enhanced under ether anesthesia (such as neomycin as reported) and was antagonized by neostigmine.
[Medicine for pregnant and lactating women]
1. This product can pass through the placenta tissue. Animal experiments have teratogenic effects. This product is forbidden for pregnant women.
2. This product is forbidden for breastfeeding women, and breastfeeding should be stopped when it is clearly indicated.
【Children's Medication】
It is not clear, but it is not recommended to use this product in pediatric patients.
【Medications for the Elderly】
The clinical data of this product is not enough to explain the safety and effectiveness of patients 65 years and older to determine whether they and young patients have different reactions to this product. Elderly patients usually start with the lowest dose. It is known that capreomycin sulfate is basically excreted by the kidneys, so the risk of drug toxicity may be greater in patients with impaired renal function. As the renal function of the elderly is physiologically diminished, the dose needs to be adjusted according to the renal function, and monitoring of renal function may be useful.
The elderly are also more likely to suffer from hearing impairment. Before starting treatment with this product, hearing and vestibular function tests should be performed, and regular tests should be performed during the treatment.
【medicine interactions】
1. Combined use with aminoglycosides can increase the possibility of ototoxicity, nephrotoxicity and neuromuscular blockade, and hearing loss, which may be temporary, may continue to progress to deafness after stopping the drug, which is often permanent. Neuromuscular blockade can cause skeletal muscle weakness and respiratory depression or respiratory muscle paralysis. The use of anticholinesterase drugs or calcium salts can help restore.
2. Simultaneous or sequential application with amphotericin B, vancomycin, bacitracin, paromomycin, cyclosporine, carmustine, cisplatin, bumetanide, ethanilic acid, furosemide can increase ototoxicity As well as the possibility of nephrotoxicity, hearing and renal function tests should be performed regularly when used together.
3. Antihistamines, buclizine, Cyclizine, meclizine, phenothiazines, thioxanthenes, trimebenzamide, when used in combination with this product may conceal tinnitus, dizziness or dizziness and other ototoxic symptoms.
4. When combined with anti-neuromuscular blockers, it can antagonize the effect of the latter on skeletal muscle. Therefore, at the time of combined use or after combined use, the dose of the anti-muscle asthenia drug needs to be adjusted.
5. When methoxyflurane or polymyxin injections are used simultaneously or sequentially with this product, nephrotoxicity or neuromuscular blockade may increase, so it should be avoided in combination; neuromuscular blockade can cause skeletal muscle weakness and respiratory depression or Respiratory muscle paralysis; caution should also be exercised when this product is used in combination with both during or after surgery. The use of anticholinesterase drugs or calcium salts can help block recovery.
6. When this product is used in combination with opioid analgesics, the central respiratory depression effects of the two may add up, resulting in aggravated respiratory depression or prolonged inhibition time or respiratory paralysis. Close observation and follow-up are necessary.
【Overdose】
Symptoms and signs
The nephrotoxicity caused by the injection of this product is closely related to the area under the blood concentration-time curve. Elderly patients, patients with abnormal renal function or dehydration, and patients receiving other nephrotoxic drugs at the same time have a higher risk of acute tubular necrosis.
The patient's hearing and the 8th pair of cranial nerve vestibular damage are related to their own renal dysfunction or dehydration, receiving treatment with this product and receiving other additional toxic drugs. Patients may present with dizziness, tinnitus, vertigo, and loss of high hearing acuity.
Neuromuscular blockage or respiratory paralysis may occur after rapid intravenous infusion.
It is reported in the literature that patients with overdose of this product can manifest as hypokalemia, hypocalcemia, hypomagnesemia, and electrolyte disorders similar to Bartter´s syndrome.
treatment
Unblock the patient's airway. Closely monitor the patient's vital signs, blood gas, body fluid balance, electrolytes and renal function. Consider using activated charcoal to replace or assist gastric emptying. In many cases, activated charcoal is more effective than vomiting or lavage. Repeated use may speed up the removal of absorbed drugs. When using gastric emptying or activated charcoal, care should be taken to keep the patient’s airway open. Patients with renal insufficiency should pay attention to hydration to maintain the urine output at 3~5mL/kg/h.
For patients with obvious renal disease, hemodialysis is used to remove this product.
【Pharmacology and Toxicology】
Pharmacological action
Capreomycin has antibacterial effect on human Mycobacterium tuberculosis. The antibacterial mechanism of capreomycin is not clear. Because it is similar in structure to violomycin (VM), it has cross-resistance. It is speculated that its mechanism of action is the same as that of aminoglycosides. It binds to the ribosome of tuberculosis and affects the synthesis of bacterial protein, resulting in antibacterial and bactericidal effects.
In vitro antibacterial test: According to domestic reports, the minimum inhibitory concentration for tuberculosis H37RV strain is 3.1~4.0μg/ml, and some are as low as 1.6μg/ml. Foreign reports are 3.13~6.25μg/ml. With >10μg/ml as the limit of resistance, there are very few people who are resistant to capreomycin in some domestic investigations, only about 0.8% to 2.4%. Those who are resistant to streptomycin are all sensitive to capreomycin. Capreomycin is generally considered to be one of the slower anti-tuberculosis drugs, but it is still easy to resist when used alone. The in vitro bacteriostatic test showed that capreomycin has a good bacteriostatic effect on some gram-positive cocci such as Streptococcus pneumoniae and Streptococcus b, and it is effective on some gram-negative bacilli, mainly pneumoniae. It has a strong effect on cocci.
Toxicology Research
Nephrotoxicity and eighth brain neurotoxicity can be seen in animal toxicity studies. In addition, cataracts occurred in 2 dogs in the 62mg/kg and 100mg/kg dose groups in the delayed dosing of dogs. In reproductive toxicity studies, at a dose of 50 mg/kg or higher per day, corrugated ribs with a low incidence can be seen in female rat pups.
【Pharmacokinetics】
This product is rarely absorbed through the gastrointestinal tract and must be injected.
The blood concentration of normal people reaches the peak value 1 to 2 hours after intramuscular injection of 1 g, with an average of 28 to 32 µg/ml. Capreomycin is excreted in the form of the original drug. The half-life is 3-6 hours. Within 6 hours, the average concentration in urine is 1.68mg/ml (average urine volume is 228ml). The inhibitory concentration can be reached in 8 small fashions. It is mainly excreted in urine, and can be excreted in the urine by 50-60% in 12 hours. The blood concentration is low at the 24th hour. Someone measured the urine concentration of 2000 µg/ml 3 hours after intramuscular injection of 1 g, and it was still 320 µg/ml 12 hours later. However, when subjects with normal renal function were given 1 g capreomycin daily for 30 days or longer, the subjects did not produce significant accumulation. In patients with renal impairment, T1/2 are prolonged, and capreomycin may accumulate in the serum.
For intravenous infusion, add 250ml of physiological saline to 1g, 60 drops/min, and finish the drip in 1 hour and 10 minutes, and measure the blood concentration at 1, 2, 3, 4, and 6 hours after instillation. It reaches 124~302µg/ml in 2 hours, which is more than 3 times that of intramuscular injection, and drops to 0 after 4 hours. The blood concentration maintenance time is shorter than that of intramuscular injection.
【Storage】 Sealed and stored in a cool and dry place (not exceeding 20℃).
[Packing] Soda-lime glass molded injection bottle, 1 bottle/box; 10 bottles/box.
[Validity] 24 months
【Executive standard】 YBH03092018
[Approval Number] (1) 0.75g (750,000 units) National Medicine Standard H20183426 (2) 1.0g (1 million units) National Medicine Standard H20183427
【manufacturer】
Company Name: Ruiyang Pharmaceutical Co., Ltd.
Production address: No. 1, Ruiyang Road, Yiyuan County, Shandong Province
Postal Code: 256100
Phone number: 4006 123458; 15853312365
Fax number: 0533-3248777
Website: http://www.reyoung.com
Mailbox: reyoung@reyoung.com